Halogen Family: Group VIIA in the periodic table; fluorine, chlorine, bromine, iodine, and astatine.
Hard Water: Water with dissolved Mg2+, Ca2+, Fe2+, or Fe3+ ions at a concentration (above 25 mg/liter) high enough to interfere with the use of soap.
Heat Capacity: The quantity of heat needed to raise the temperature of an object by 1 °C. Its units are cal/°C or J/°C, where °C = the change in temperature.
Heat of Combustion, Standard,
 H°combustion: The
enthalpy of combustion for 1 mole of a compound under standard conditions.
H°combustion: The
enthalpy of combustion for 1 mole of a compound under standard conditions.Heat of Formation, Standard,
 H°f: The enthalpy change for
the formation of 1 mole of a compound from its elements, all in their standard
states.
H°f: The enthalpy change for
the formation of 1 mole of a compound from its elements, all in their standard
states.Heat of Reaction, Standard,
 H°: The value of
H°: The value of  H for a reaction conducted so as
to maintain the pressure at 1 atm and the temperature at 25 °C.
H for a reaction conducted so as
to maintain the pressure at 1 atm and the temperature at 25 °C.Henderson-Hasselbalch Equation: pH = pKa + log ([anion]/[acid]).
Henry's Law: See Pressure-Solubility Law.
Hertz (Hz): 1 Hz = 1 cycle/second; the SI unit of frequency.
Hess Law Equation: For the change: aA + bB + ... => nN + mM + ...,
 H° = [n
H° = [n H°f(N) + m
H°f(N) + m H°f(M) + ...] -
[a
H°f(M) + ...] -
[a H°f(A) + b
H°f(A) + b H°f(B) +
...].
H°f(B) +
...].Heterocyclic Compound: An organic ring compound in which one (or more) or the ring atoms is from a nonmetal other than carbon.
Heterogeneous: Two or more phases with different properties.
Heterogeneous Catalyst: A catalyst that is in a different phase than the reactants. The reactants are adsorbed on the the catalytic surface where the reaaction occurs.
Heterogeneous Reaction: A reaction in which all of the reactants and products are in the same phase.
Hund's Rule: The lowest energy electron configuration results when electrons that occupy orbitals of equal energy are spread out over the orbitals as much as possible with spins unpaired.
Hybrid Atomic Orbitals: Orbitals formed by mixing the basic atomic orbitals of an atom. They are more effective at overlap than ordinary atomic orbitals.
Hydrate: A compound holding molecules of water usually in a stoichiometric ratio.
Hydration: The development of a solvent cage of water molecules about polar molecules or ions disolved in water.
Hydride: (1) A binary compound of hydrogen. (2) A compound containing the H- (hydride) ion.
Hydrocarbon: A compound whose molecules consists entirely of carbon and hydrogen.
Hydrogen Bond: An extra strong dipole-dipole attraction that occurs between molecules in which hydrogen is covalently bonded to nitrogen, oxygen, or fluorine.
Hydrogen Electrode: The standard of comparison for reduction portentials, 2H+(aq) + 2e- <=> H2(g), E°H+ = 0.00 V at 25 °C, 1 atm, and 1 M H +.
Hydrolysis of Salts: The reaction of a salt with water to affect the pH of the solution.
Hydronium Ion: H3O+.
Hydrophilic Group: Any polar organic group that cannot be involved in hydrogen bonds with water.
Hypertonic Solution: A solution whose osmotic pressure is greater than a reference solution.
Hypothesis: Tenetative explanation of the results of experiments.
Hypotonic Solution: A solution whose osmotic pressure is less than a reference solution.
Ideal Gas: A hypothetical gas that obeys the gas laws exactly.
Ideal Gas Law: PV = nRT.
Ideal Solution: The hypothetical solution that would obey the vapor pressure-concentration law (Raoult's law) exactly.
Induced Dipole: A dipole created when the electron cloud of an atom or a molecule is distorted by a neighboring dipole or by an ion.
Inert Gases: The noble gases.
Initiation Step: The first step in a chain reaction in which a reactant molecule is converted to one or more free radicals.
Initiator: A substance that, like a catalyst, promotes a reaction, but is used up during the reaction.
Inner Transition Elements: The two long rows of elements below the main body of the periodic table. They include elements 58-71 and 90-103.
Instantaneous Dipole: A momentary dipole caused by the erratic movement of electrons.
Intermolecular Attractions: Attractions between neighboring molecules.
Inverse Square Law: In the science of atomic radiations, the relationship between radiation intensity and distance: radiation intensity
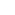 1/d2.
1/d2.Ion: An electrically charged particle.
Ion Product: A product of ion concentrations, each of which is raised to a power that is equal to the number of ions of that kind obtained from one formula unit of the salt. The mass action expression for a solubility equilibrium.
Ion-Electron Method: A method for balancing redox reactions that uses half-reactions.
Ionic Bond: The attractions between ions that hold them together in ionic compounds.
Ionic Compound:A compound composed of positive and negative ions.
Ionic Crystal: A crystal that has ions located at the lattice points.
Ionization Energy (IE): The energy needed to remove an electron from an isolated gaseous atom, ion, or molecule (usually expressed in units of kJ/mol).
Ionizing Radiation: Radiation arising from radioactivity or other nuclear reactions that generates ions as it passes through matter.
Ion-Product Constant of Water: [H+][OH-] = Kw.
Isomer: ONe of a set of compounds having identical molecular formulas but different structures.
Isomerism: The phenomenonof the existence of sets of compounds having the same moelcular formular but different structures.
Isotonic Solution: A solution whose concentration is such that it has the same osmotic pressure as a reference solution.
Isotope Dilution Analysis: An analytic technique for measuring volumes by studying the extent to which the specific activity of a sample of radioactive material is reduced by its being biluted into the larger volume.
Isotopes: Atoms of the same element having different mass numbers caused by differences in their numbers of neutrons.
IUPAC Rules: The formal rules for naming compounds as developed by the International Union of Pure and Applied Chemisty.
Joule (J): The SI unit of energy. 1 J = 1 kg m2/s2; 4.184 J = 1 cal.
Ka: See Acid Ionization Constant.
Kb: See Base Ionization Constant.
Kw: See Ion-Product Constant of Water.
Kelvin Scale: Absolute temperature scale having degree units called kelvins (K). Degree size is the same as the Celsius degree. K = °C + 273.15.
Ketone: An organic compound with the
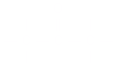 group.
group.Kilogram (kg): 1 kg = 1000 g; the base unit for mass in the SI.
Kinetic Energy: Energy of motion: KE = 1/2mv2
Kinetic Theory of Gases: A set of postulates about gases used to explain the gas laws. A gas consists of an extremely large number of very tiny, hard particles in constant random motion. They have negligible volume and, between collisions, they mov e in straight lines.
Lanthanide Contraction: The decrease in siz experienced by elements 58-71 that causes the elements that follow the lanthanides to have unusually small sizes.
Lanthanide Elements: Elements 58-71.
Lattice Energy: Energy released by the imaginary process in which isolated ions come together to form a crystal of an ionic compound.
Law: Statement of behavior based on the results of many experiments. Laws do not offer explanations of behavior.
Law of Conservation of Mas: Mass is neither gained nor lost in a chemical change; mass is conserved.
Law of Constant Heat Summation (Hess's Law): For any reaction that can be written in steps, the standard heat of reaction is the sum of the standard heats of reaction for the steps.
Law of Definite Proportions: In a given chemical compound, the elements are always combined in the same proportion by mass.
Law of Multiple Proportions: Whenever two elements form more than one compound, the different masses of one that combines with the same mass of the other are in the ratio of small whole numbers.
Le Châtelier's Principle:
Lewis Acid: An electron-pair acceptor.
Lewis Base: An electron-pair donor.
Lewis Structure: Also called Lewis formula. A structural formula drawn with Lewis symbols that shows the valence electrons using dots and dashes.
Lewis Symbol: The symbol of an element surrounded by dots that represent the valence electrons of an atom of that element.
Ligand: A molecule or anion that can bind to a metal ion to form a complex.
Lipid: Any substance found in plants or animals that can be dissolved in nonpolar solvents.
Liquid Crystal: A substance that is able to flow like a liquid but that has some physical properties normally associated with crystals.
Liter: 1 liter = 1 dm3 = 1000 cm3 = 1000 ml.
London Forces: Weak attractive forces caused by instantaneous dipole-induced dipole attractions.
Lone Pair: A pair of electrons in the valence shell of an atom that is not shared with another atom. an unshared pair of electrons.
M-O
Macromolecule: A molecule with hundreds to thousands of atoms such as found in polymers.Magic Numbers: In nuclear chemistry, the numbers 2, 8, 20, 50, 82, and 126, and whose significance is that nuclides are expected to be particularly stable when the number of protons or neutrons (and especially both) equals on of these numbers.
Magnetic Quantum Number: m, which can have integer values from -l to +l.
Malleability: A metal's ability to be hammered or rolled into thin sheets.
Manganese Nodules: Lumps the size of an orange that contain large concentrations of manganese and iron. They are found on the ocean floor.
Mass: A measure of the amount of matter that there is in a given sample.
Mass Action Expression: A fraction in which the numerator is the product of the molar concentrations of the producets raised to powers equal to their coefficients and the denominator is the product of the molar concentrations of the reactants raise d to powers equal to their coefficients. For gaseous reactions, parital pressures can be used in place of molar concentrations.
Mass Number: The numerical sum of the protons and neutrons in an atom of a given isotope.
Matter: Anything that has mass and occupies space.
Mechanism: The series of individual steps in a chemical reaction that gives the net overall change.
Metallic Crystal: A solid having positive ions at the lattice positions that are attracted to a "sea of electrons" that extends throughout the entire crystal.
Metalloids: Elements with properties that lie between those of metals and nonmetals. They are located around the diagonal line running from boron (B) to astatine (At) in the periodic table.
Metallurgy: The science and technology of metals; it is concerned with the procedures and chemical reactions that are used to separate metals from their ores and make them ready for pratical uses.
Metathesis Reaction: See Double Replacement Reaction.
Meter (m): SI base unit for length.
Metric System: A decimal system of units.
Milliliter (ml): 0.001 liter = 1 ml; 1000 ml = 1 liter.
Millimeter (mm): 0.001 m = 1 mm; 1000 mm = 1 m.
Millimeter of Mercury (mm Hg): A unit of presure equal to 1/760 atm. 760 mm Hg = 1 atm.
Mineral: A specific solid in the earth's crust having a more or less definite chemical formula.
Mixture: Any matter consisting of two or more substances physically combined in no particular proportion by mass.
Model, Scientific: A picture or a mental construction derived from a set of ideas and assumptions that we imagine to be true because they enable us to explain certain observations and measurements (e.g., the model of an ideal gass; Bohr's atomic mo del).
Molal Concentration (m): The number of moles of solute in 1000 g of solvent; the molality of a solution.
Molality: See Molal Concentration.
Molar Concentration (M): The number of moles of solute per liter of solution.
Molar Heat of Fusion: Heat absorbed when 1 mol of a solid melts to give 1 mol of liquid at constant temperature and pressure.
Molar Heat of Sublimation: Heat absorbed when 1 mol of solid sublimes to give 1 mol of vapor at constant temperature and pressure.
Molar Heat of Vaporization: Heat absorbed when 1 mol of liquid is converted to 1 mol of vapor at constant tmemperature and pressure.
Molar Mass: The number of grams per mole of a compound or element.
Molar Solubility: The number of moles of solute required to give 1 liter of a saturated solution of the solute.
Molar Volume, Standard: The volume of 1 mol of gas at STP; 22.4 liter/mol.
Molarity: See Molar Concentration.
Mole (mol): The SI unit for amount of substance; the formula weight in grams; a quantity of chemical substance containing 6.02 x 1023 formula units.
Mole Fraction: The ratio of the number of moles of one component of a mixture to the total number of moles of all components.
Mole Percent: The mole fraction of a component expressed as a percent.
Molecular Crystal: A crystal that has molecules or individual atoms at the lattice points.
Molecular Equation: A chemical equation giving the full formulas of all the reactants and products that will be involved in an actual experiment.
Molecular Formula: A chemical formula that gives the actual composition of one molecule.
Molecular Orbital Theory: Theory of covalent bonding that views a molecule as a collection of positive nuclei surrounded by electrons distributed among a set of bonding and antibonding molecular orbitals of different energies.
Molecular Orbitals: Orbitals that extend over two or more atomic nuclei.
Molecular Weight: See Formula Weight.
Molecule: A neutral particle composed of two or more atoms.
Monodentate Ligand: A ligand that can attach itself to a metal ion by only one atom.
Monomer: A substance of low formula weight used to make a polymer.
Monoprotic Acid: An acid that can furnish one H+ per molecule.
Monosaccharide: A carbohydrate that cannot be hydrolyzed.
Nematic Liquid Crystal: Composed of long, rodlike molecules packed like short pieces of uncooked spaghetti.
Nernst Equation: Ecell = E°cell - 0.0592/n x log Q.
Net Ionic Equation: A chemical equation having no spectator ions or molecules and that satisfies both a material and an electrical balance.
Neutral Solution: A solution in which [H+] = [OH-].
Neutralization, Acid-Base: The destruction of an acid by a base or of a base by an acid.
Neutron: A subatomic particle with a charge of zero and a mass of 1.008665 amu (1.674954 x 10-24 g) that exists in all atomic nuclei except those of the hydrogen-1 isotope, its symbol is n or 10n in nu clear equations.
Neutron Activation Analysis: A technique for analyzinag for trace impurities by rendering them radioactive through neutron bombardment and then studying the frequencies and intensities of the gamma radiations they emit.
Neutron Emission: A nuclear reaction in which a neutron is ejected.
Nitrogen Family: Gourp VA in the periodic table; nitrogen, phosphorus, arsenic, antimony, and bismuth.
Nitrogen Fixation: The conversion of atmospheric nitrogen into chemical forms used by plants and accomplished soil microorganisms with the assistance of plants.
Noble Gases: Elements of group 0 in the periodic table; helium, neon, argon, krypton, xenon, radon.
Node: A place where the amplitude or intensity of a wave is zero.
Nonelectrolyte: A compound that in its molten state or in solution will not conduct electricity.
Nonstoichiometric Compound: A substance in which the elements are combined in a not quite whole number ratio by moles.
Nonvolatile: Describing a substance with a high boiling point, a low vapor pressure, and that does not evaporate.
Normal Boiling Point: The temperature at which the vapor pressure of a liquid equals 1 atm.
Normality (N): The number of equivalents per liter of an acid or a base or other reactant.
Nuclear Equation: A shorthand expression of a nuclear reaction that is balanced when the sums of the atomic numbers on either side of the arrow are equal and the sums of the mass numbers are equal.
Nucleic Acids: Polymers in plants and animals that are involved in the chemistry of heredity and whose molecules when hydrolyzed, give a sugar unit, a phospate ion, and a set of four or five nitrogen-containing, basic hetercyclic compounds.
Nucleon: A general name for protons or neutrons.
Nucleus: The hard, dense core at the center of an atom that contains all of the atom's protons and neutrons.
Octet Rule: An atom tends to gain or lose electrons its outer shell consists of eight electrons.
Odd-Even Rule: In nuclear chemistry, a rule that says when the numbers of protons and neutrons in a nucleus are both even, the isotope is far more likely to be stable that when both even, the isotope is far more likely to be stable that when both n umbers are odd.
Open Hearth Furnace: A furnace used to convert pig iron into steel.
Optical Isomers: Stereo isomers other that geometric isomers.
Orbital: A particular electron waveform with a particluar energy. In an atom, each orbital has s pecific set of values for (n), (l), and (m).
Orbital Diagram: A diagram showing an atom's orbitals in which the electrons are represented by arrows to indicate paired and unpaired spins.
Order (of Reaction): The sum of the exponents in the rate law is the overall order of the reaction. Each exponent gives the order with respect to a specific reactant.
Ore: A mineral that can be extracted from the earth's crust and processed at a profit.
Organic Compound: Any compound of carbon other that a carbonate, bicarbonate, cyanide, cyanate, carbide or gaseous oxide.
Osmolality: The molal concentration of all particles of solutes--molecules or separated ions--that contribute to the osmotic pressure of a solution.
Osmolarity: The molar concentration of all particles of solutes--molecules or separated ions--that contribute to the osmotic pressure of a solution.
Osmosis: The passage of solbent molecules, but not those of solutes, through a semipermeable membrane; the limiting case of dialysis.
Ostwald Process: An industrial synthesis of nitric acid from ammonia.
Overlap: A portion of two orbitals from different atoms sharing the same space.
Oxidation: Loss os electrons; increase in oxidation number.
Oxidation Number: The charge an atom in a compound would have if all of the electrons in the bonds belonged entirely to the more electronegative atoms.
Oxidation State: Means the same as oxidation number.
Oxidation-Reduction Reaction: A reaction involving the transfer of electrons from one species to another.
Oxidizing Agent: Substance that causes oxidation; it is reduced
Oxoacid: An acid containing hydrogen, oxygen, and another element. (e.g., HNO3, H3PO4, H2SO4).
Oxygen Family: Group VIA in the periodic table: oxygen, sulfur, selenium, tellurium and polonium.
P-R
Paramagnetism: Weak magnetism that a substance has when it contains unpaired electrons.Partial Charge: Charges at opposite ends of a dipole that are less than 1 + or 1 - charges.
Partial Pressure: The pressure contributed by an individual gas in a mixture of gases.
Partial Pressures, Law of (Dalton's Law of Partial Pressures):The total pressure of a mixture of gases equals the sum of their partial pressures. Pt = Pa + Pb + Pc + . . .
Pascal (Pa): The SI unit of pressure; 133.3224 Pa = 1 torr.
Pauli Exclusion Principle: No two electrons in the same atom can have the same values for all four of their quantum numbers.
Peptide Bond: The amide linkage in protein chemistry:
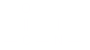
Percent Concentration:
Weight/weight (w/w) |
The grams of solute in 100g of solution |
Volume/volume (v/v) |
The number of volumes of solute in 100 v of solution. |
Weight/volume (w/v) |
The grams of solute in 100 ml of solution. |
Milligram percent (mg %) |
The milligrams of solute in 100 ml of solution. |
Percentage by Weight: The number of grams iof an element combined in 100 g of a compound.
Percentage Compostion: A complete list of the percentages by weight of a compounds elements.
Period: Horizontal row of elements in the periodic table.
Peroxide: A compound whose molecules have two oxygen atoms joined by a single bond.
pH: pH = -log [H+].
Phase: A homogeneous region within a sample.
Phase Diagram: A pressure-temperature graph on which are plotted temperatures and pressures at which equilibrium exists between the states of a substance. It defined T - P regions in which the solid, liquid and gaseous states of the substance can exist.
Photon: "Packet" of energy in electromagnetic radiation; its energy, E = h
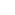 where
where 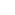 is the frequency and h is Planck's
constant.
is the frequency and h is Planck's
constant.Photosynthesis: The use of solar energy by a plant to synthesize high-energy molecules from simpler, low-energy molecules of CO2 and H2O. The primary products are carbohydrates. The plant's chlorophyll is the solar energy ab osorber.
Physical Property: Property that can be specified without reference to another substance, for example, color or volume.
Pi Bond (
 Bond): A bond formed by the sideways overlap of a pair of p orbitals.
Electron density is concentrated in two separate regions that lie on opposite
sides of the imaginary line joining the nuclei.
Bond): A bond formed by the sideways overlap of a pair of p orbitals.
Electron density is concentrated in two separate regions that lie on opposite
sides of the imaginary line joining the nuclei. Pig Iron: The impure iron that comes from the blast furnace.
pKa: pKa = -log Ka.
pKb: pKb = -log Kb.
Plastic: A finished or semifinished article made by molding, casting, extruding, drawing, or laminating a resin.
Plasticizer: An oily organic material added to a resin to reduce hardness and brittleness in the final plastic article.
pOH: pOH = -log [OH-].
Polar Covalent Bond (Polar Bond): A covalent bond in which more that half of the bon's negativ charge is concentrated around one of the two atoms.
Polyatomic Ion: An ion composed of two or more atoms.
Poludentate Ligand: A ligand that has two or more atoms that can become simutaneously attached to a metal ion.
Polymer: A substance consisting of macromulecules having repeating structural units.
Polymerization: A chemical reaction that converts a monomer into a polymer.
Polyolefin: A polymer of any member of the alkene ("olefin") family.
Polypeptide: A polymer of
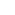 -amino acids that makes up all or most of a
protein.
-amino acids that makes up all or most of a
protein.Polyprotic Acid: An acid that can furnish more that one H+ per molecule.
Polysaccharide: A carbohydrate whose molecules can by hydrolyzed to hundreds of monosaccharide molecules.
Positron: A positively charged particle with the mass of an electron. Its symbol if 01p.
Post-Transition Metal: A metal that occurs in the periodic table immediately
Potential Energy: Stored energy
Precipitate: A solid that separates from a solution usually as the result of a chemical reaction.
Precipitation: In chemistry, the formation of a solid that separates from a solution usually as the result of a chemical reaction.
Precision: How reproducible measurements are.
Pressure-Solubility Law (Henry's Law): The concentration of a gas dissolved in a liquid at any given temperature is directly proportional to the partial pressure of that gas on the solution.
Pressure-Temperature Law (Gay-Lussac's Law): The pressure of a given mass of gas is directly proportional to its Kelvin temperature if the volume is kept constant. P1/T1 = P2/T2
Pressure-Volume Law (Boyle's Law): The volume of a given mass of gas is inversely proportional to its pressure (at constant temperature.) P1V1 = P2V2.
Principal Quantum Number: n, which can have values of 1, 2, 3, . . . ,
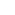 .
.Products: Substances whose formulas appear on the right side of the arrow in a chemical equation. The substances produced in a chemical reaction.
Propagation Step: A step in a chain reaction in which a free radical reacts with a reactant molecule to give a product molecule and another free radical.
Property: A characteristic of matter.
Protein: A macromolecular substance found in nature consisting wholly or mostly of one or more polypeptides often combined with an organic molecule and sometimes also combined with a metal ion.
Proton: (1) A subatomic particle with a charge of 1 + and a mass of 1.007276 amu (1.672649 x 10-24 g) that exists in a atomic nuclei. (2) The name often used for the hydrogen ion and symbolized as H+ in acid-base discussions a nd either 11p or 11H in discussions of tranmutations.
Pure Substance: Element or compound.
Quantized Energy: Discrete amount of energy.
Quantun: The energy of one photon.
Quantum Mechanics: See Wave Mechanics.
Quantum Number: A number realted to the energy, shape, or orientation of an orbital. Also, a number related to the spin of the electron.
R: See Gas Constant, Universal.
Rad (rd): A unit of radiation-absorbed-dose. 1 rd = 10-5 J/g = 10-2 Gy.
Radioactive Decay: The change of a nucleus into another nucleus (or a more stable form) by the loss of an alpha or beta particle or a gamma ray photon.
Radioactive Disintegration Series: A sequence of nuclear reactions beginning with an unstable actinide and eding with a stable isotope of lower atomic number.
Radioactivity: The emission of one or more kinds of radioation from an isotope with unstable nuclei.
Radiological Dating: A technique for measuring the age of some geologic formation or ancient artifact by measuring the ratio of the concentrations of two isotopes, once radioactive and the other a stable decay product.
Radionuclide: A radioactive isotope.
Raoult's Law: See Vapor Pressure-Concentration Law.
Rare Earth Metals: The lathanides.
Rate: A ration in which units of time appear in the denominator, for example, 40 miles/hr or 3.0 mol/liter/s.
Rate Constant: The proportionality constant in the rate law; the rate of reaction when all reactant concentrations are 1 M.
Rate of Reaction: How quickly the reactants disappear and the products form, expressed in units of mol liter-1 s-1.
Rate Law: An equation that relates the rate of a reaction to the molar concentrations of the reactants raised to powers.
Rate-Determining Step (Rate Limiting Step): The slow step in a reaction mechanism.
Reactant LimitingThe reactant in a given reaction that is present in a deficient quantity according to the stoichiometry of the reaction.
Reactants: Substances whose formulas appear on the left side of the arrow in a chemical equation. The substances brought together to react.
Reaction Quotient: See Mass Action Expression.
Reagent: Any chemical, mixture of chemicals, or solution of chemicals used as part of an experimental reaction.
Redox Reaction: Oxidation-reduction reaction.
Reducing Agent: Substance that causes reduction; it is oxidized.
Reduction: Gain of electrons; decrease in oxidation number.
Reduction Potential: A measure of the tendency of a given half-reaction to occur as a reduction.
Rem: A dose in rads multiplied by a factor that takes into accoun the variations that different radiations have in their damage-causing abilities in tissue.
Replication: In nucleic acid chemistry, the reporductive duplication of DNA double helixes prior to cell division.
Representative Element: An element in one of the A-groups in the peridic table.
Resin: A raw, unfabricated polymer used to make all or most of a plastic article.
Resonance: A concept in which the actual Lewis structure of a molecule of polyatomic ion is represented as a composite or average of two or more resonance structures. The individual resonance structures themselves do not actually exist.
Resonance Hybrid: The actual structure of a molecule or polyatomic ion that is represented by two or more resonance structures.
Reversible Process: A process that occurs by an infinite number of steps during which the driving force for the change is just barely greater that the force that resists the change.
Ring Compound: A compound whose molecules include closed-chain sequences of atoms.
RNA: Ribonucleic acid; nucleic acid thatm when hydrolyzed, gives ribose, phosphate ion, and chiefly four hetercyclic amines--adenine, uracil, guanine, and cytosine. Varieties include:
mRNA |
or messenger RNA made at the direction of DNA; and the carrier of genetic message (as a series of codons) from the cell nucleus to the assembly site for making polypeptides. |
tRNA |
or transfer RNA--amino acid carrier RNA. |
Roasting: Heating an ore in air, which converts sulfides to oxides.
Rock: A complex mixture of minerals.
S-T
Salt Bridge: A tube containing an electrolyte that connects the two half-cells of a galvanic cells.Saturated Organic Compound: Any organic compound having only single bonds.
Saturated Solution: A solution in which there is an equilibrium between the dissolved and undissolved states of the solute.
Scientific Method: Observation, explanation, and testing of an explanation by additional experiments.
Scientific Notation: Numbers represented as a decimal number between 1 and 10 multiplied by 10 raised to a power; for example, 6.02 x 1023
Second Law of Thermodynamics: Whenever a spontaneous event takes place it is accompanied by an increase in the entropy of the universe.
Secondary Quantum Number: l, which can have values of 0, 1, . . . , (n - 1).
Semiconductor: A substance that conducts electricity weakly.
Shell: All orbitals of a given n.
Sigma Bond (
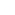 Bond): A bond formed by the
"head-to-head" overlap of two orbitals. The electron density is concentrated
along the imaginary line joining the two nuclei.
Bond): A bond formed by the
"head-to-head" overlap of two orbitals. The electron density is concentrated
along the imaginary line joining the two nuclei.Significant Figures: In a measured quantity, the number of digits known for sure plus the first one that is uncertain.
Simple Cubic Unit Cell: A cubic unit cell with atoms, molecules, or ions only at the corners.
Single Bond: A covalent bond in which a single pair of electrons are shared.
Slag: A relatively low melting mixture of impurities that forms in the blast furnace and other furnaces used in refining metals.
Smectic Liquid Crystals: Rodlike molecules in layers of parallel rods.
Sol: The colloidal dispersion of a solid in a fluid.
Solubility: The ratio of solute to solvent in a saturated solution; usually expressed as the number of grams of solute per 100 g of solvent at a specified temperature.
Solubility Product Constant, Ksp: The equilibrium constant for the solubility of a salt. For a saturated solution, Ksp is equal to the product of the molar concentrations of the ions, each raised to an appropria te exponent.
Solute: Something dissolved in a solvent to make a solution.
Solution: A homogeneours mixture in which all particles are the size of atoms, small molecules or small ions.
Solvation: The development of a cagelike network of solvent molecules about a molecule or ion of the solute.
Solvay Process: Used to prepare Na2CO3 from NaCl, CO2 and NH3.
Solvent: A medium, usually a liquid, into which something (the solute) is dissolved to make a solution.
sp Hybrid Orbitals: Hybrid orbitals formed by mixing one (s) and one (p) atomic orbitals. The angle between a pair of sp hybrids is 180°
sp2 Hybrid Orbitals: Hybrid orbitals formed by mixing one (s) and two (p) atomic orbitals. The angle between any two sp2 hybrids is 120°
sp3 Hybrid Orbitals: Hybrid orbitals formed by mixing one (s) and three (p) atomic orbitals. The angle between any two sp3 hybrids is 120°.
Specific Heat: The quantity of heat that will raise the temperature of 1 g of a substance by 1 °C. (Its units are cal/g °C, where °C = the change in temperature.)
Spectrochemical Series: A listing of ligands in order of their ability to produce a large crystal field splitting.
Spin Quantum Number: ms, which can have values of +1/2 and -1/2.
Spontaneous Change: A change that occurs by itself without outside assistance.
Standard Cell Potential: The potential of a galvanic cell at 25 degrees celsius and 1 atm when all ionic concentrations are exactly 1 M.
Standard Conditions of Temperature and Pressure (STP): 273 K (0 °C) and 760 Torr.
Standard Enthropy, S°: The entropy of 1 mol of a substance has at 25 °C and 1 atm.
Standard Entropy Change,
 S°:
S°:  S° = (Sum of S° of products) - (Sum of S° of
reactants).
S° = (Sum of S° of products) - (Sum of S° of
reactants).Standard Free Energy Change,
 G°:
G°:  G° =
G° =  H° - T
H° - T S°
S°Standard Reduction Potential: The reduction potential of a half-reaction at 25 °C and 1 atm when all ion concentrations are 1 M.
Standard Solution: Any solution whose concentration is accurately known.
Standard State: The condition in which a substance is in its most stable form at 25 °C and 1 atm.
Standing Wave: A wave whose peaks and nodes do not change position.
State: Solid, liquid, or gas. In thermodynamics, a particular set of conditions of temperature, composition, and pressure.
State Function: A thermodynamic function whose value depends only on the initial and final states of the system and not on the path used to get from the initial to the final state. P, V, T, H, S, and G are all state functions.
State of a System: The specified values of the physical properties of a system--physical form, composition, concentration, temperature, and pressure.
Stereoisomers: Isomers whose structures differ only in spatial orientations (geometric iosmers; optical isomers).
Stock System: System of nomenclature that uses Roman numerals to specify oxidation states.
Stoichiometry: A description of the relative quantities by moles of the reactants and products in a reactionas given by the coefficients in the balanced equation.
STP: See Standard Conditions of Temperature and Pressure.
Straight-Chain Compound: An organic compound in whose molecules the carbon atoms are joined in one continuous open chain sequence.
Strong Acid: An acid that is essentially 100% ionized in water. A good proton donor. An acid with a very large value of Ka.
Strong Base: Any powerful proton acceptor; a base with a very large value of Kb; a metal hydroxide that ionizes almost 100% in water.
Structural Formula: A chemical formula that shows how the atoms of a molecule are arranged and to which other atoms they are bonded.
Sublimation: Conversion of a solid directly to a gas without passing through the liquid state.
Subshell: All orbitals of a given shell that have the same value of l.
Substrate: In enzyme chemistry, the substance whose reaction is catalyzed by the enzyme.
Supercooled Liquid: A liquid at a temperature below its freezing point. An amorphous solid.
Supercooling: Cooling a liquid to a temperature below its freezing point.
Supercritical Fluid: A substance at a temperature above its critical temperature.
Superimposability: A test of molecular chirality in which a model of a molecule and a model of the mirror image of this molecule are compared to see if the two can be made to blend perfectly, with every part of one coinciding simultaneously with th e parts of the other.
Superoxide: An oxide containing the O2- ion.
Supersaturated Solution: Any solution containing more solute than a saturated solution would ordinarily hold at the given temperature.
Surface Tension: A measure of the amount of energy needed to expand the surface area of a liquid.
Surfactant: A substance that lowers the surface tension of a liquid and promotes wetting.
Surroundings: That part of the universe other than the system being studied and separated from the system by a real or an imaginary boundary.
Suspension: A homogeneous mixture in which the particles of one (or more) of the substances are relatively large (10 nm in one dimension) and are dispersed uniformly in some medium.
System: That part of the universe under study and separated from the rest of the universe (the surroundings) by a real or an imaginary boundary.
t1/2: See Half-Life.
Temperature-Volume Law (Charles' Law): The volume of a given mass of gas is directly proportional to its Kelvin temperature if the pressure is kept constant. V1/T1 = V2/T2
Theory: Tested explanation of the results of many experiments.
Thermite Reaction: The very exothermic reaction, 2Al + Fe2O3 => 2Fe + Al2O3.
Thermodynamics: The study of energy changes and the flow of energy from one substance to another.
Thermometer: Device used to measure temperature.
Thermotropic Substance: A substance that behaves like a liquid crystal over a limited temperature range.
Third Law of Thermodynamics: For a pure crystalline substance, S = 0 at 0 K.
Torr: A unit of pressure equal to 1 mm Hg.
Trace Element: In enzyme chemistry, any metal or nonmetal ion needed by an enzyme.
Tracer Analysis: The use of radioactivity to trace the movement of something or to locate the site of radioactivity.
Transcription: In nucleic acid chemistry, the synthesis of mRNA at the direction of DNA.
Transition Elements: Elements in the periodic table located between Groups IIA and IIIA.
Transition Metals: Transition elements.
Transition State: The brief moment during a reaction when the reactants have collided and are at the high point on the potential energy diagram for the reaction.
Translation: In nucleic acid chemistry, the synthesis of a polypeptide at the direction of a molecule of mRNA.
Transmutation: The conversion of one isotope into another.
Transuranium Elements: Elements 93 and higher.
Traveling Wave: A wave whose peaks and nodes move.
Triple Bond: A covalent bond in which three pairs of electrons are shared.
Triple Point: The temperature and pressure at which the liquid, solid, and vapor states of a substance can coexist in equilibrium.
Triprotic Acid: An acid that can furnish three H+ per molecule.
Tritium: Hydrogen-3; 31H (sometimes 31t); an isotope of hydrogen.
Tyndall Effect: The scattering of light by colloidally dispersed particles that gives a milky appearance to the mixture.
U-Z
Uncertainty Principle: There are limits in our ability to measure a particle's speed and position simultaneously.Unit Cell: The smallest portion of a crystal that can be repeated over and over in all directions to give the crystal lattice.
Unsaturated Solution: Any solution with a concentration less than that of a saturated solution of the same solute.
Vacuum: An enclosed space containing no matter whatsoever. A partial vacuum is an enclosed space containing a gas at a very low pressure.
Valence Bond Theory: Theory of covalent bonding that views a bond as being formed by the sharing of one pair of electrons between two overlapping atomic or hybrid orbitals.
Valence Electrons: Electrons in the valence shell.
Valence Shell: In an atom, the shell with highest n.
Valence Shell Electron Pair Repulsion Theory: A theory used to predict molecular structure. It is based on the idea that electron pairs (bonding and lone pairs) in the valence shell of an atom stay as far apart as possible.
Vapor Pressure: The pressure exerted by the vapor above a liquid. If the liquid and vapor are in equilibrium, this pressure is the equilibrium vapor pressure.
Vapor Pressure-Concentration Law (Raoult's Law): The vapor pressure of one component above a mixture of molecular compounds equals the product of its vapor pressure when pure (at the same temperature) and its mole fraction.
Vitamin: An organic compound present in trace concentration in various foods that is essential for health but that cannot be made by the body and whose absence leads to a specific vitamin-deficiency disease.
Volatile: Describing a liquid that has a low boiling point, a high vapor pressure, and, therefore, evaporates easily.
Volt (V): The SI unit of electrical potential or emf. 1 V = 1 J/C
Water of Hydration: Water molecules trapped in a solid substance, usually in a stoichiometric ratio to the other material.
Wave Mechanics: Theory of atomic structure based on the wave properties of matter.
Wavelength: The distance between crests in the wavelike oscillations of electromagnetic radiations; symbol,
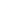 .
.Weak Acid: A poor proton donor; an acid with a value of Ka less than 10-3; any acid with a low percentage ionization in water.
Weak Base: Any poor proton acceptor; a base with a low value of Kb; any base with a low percentage ionization in water.
Weight: The force with which a substance is attracted to the earth by gravity.
Wetting: The spreading of a liquid across a surface.
X Ray: A stream of very high-energy photons emitted by substances when they are bombarded by high-energy beams of electrons or are emitted by radionuclides that have undergone K-electron capture.
Yield, Percentage: The ratio, given as a percent, of the quantity of a product actually obtained to the theoretical yield.
Yield, Theoretical: The quantity of a product calculated by the stoichiometry of the reaction.
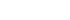 group attached
to a carbon having only single bonds.
group attached
to a carbon having only single bonds.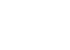 group attached to a carbon having only
single bonds.
group attached to a carbon having only
single bonds.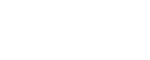 where G is a sidechain
group (or H).
where G is a sidechain
group (or H).
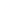 . The equation is used to analyze X-ray
diffraction data obtained from crystals.
. The equation is used to analyze X-ray
diffraction data obtained from crystals.
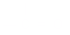
 ): One mole of electrons; 96,500
coulombs.
): One mole of electrons; 96,500
coulombs.